|
Have a grip on the fundamentals of Atomic Structure by accessing our Formulas Sheet and Tables.Ģ. You will find the concepts of Atomic Structure much simple with the formula list provided. The solid wedges indicate a bond sticking out of the plane of the screen toward you, and the dashed ones behind that plane.Formulae List of Atomic Structure makes it easy for you to evaluate the problems related in a short span of time. Is the formula for cyclohexane, shown here. It's simple and it conveys a pretty good picture of the structure, which is also bent, as shown. In this kind of notation, the ends and vertices of the stick figures all stand for carbon atoms, to which are bound all of the hydrogens that the laws of chemistry will allow (carbon can only form four bonds). 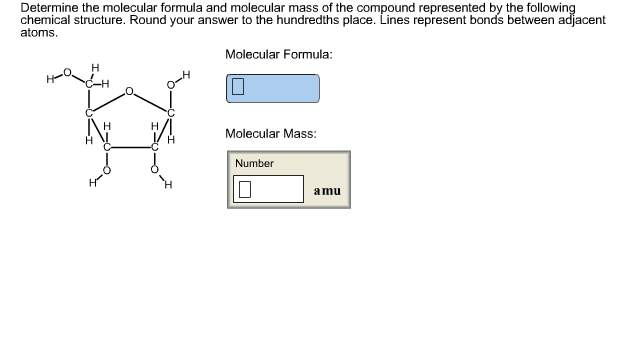
There is a shorthand notation for this kind of molecule. This one is a little better still because it shows three hydrogens bound to the left-most carbon, and the hydroxyl plus two hydrogens bound to the right C. Is a little better because it puts the OH, called a hydroxyl group, where it belongs, on the end. 
Gives us the right ratios of the atoms in this compound (ethanol), but it doesn't tell us much about what the molecule looks like. Amine groups appear in a wide variety of compounds, so you'll see them again. Here's a rough picture of the structure of cisplatin. The 2 outside the parentheses is multiplied by the implied subscript of 1 on the nitrogen, for a total of two nitrogens, and by the subscript of 3 on the hydrogen for a total of six hydrogens. The NH 3 in parentheses, known as an amine group, occurs twice. Here's how we count the atoms in that formula. Cisplatin is an important drug used in chemotherapy, and it's written like this: In such a formula, the subscript outside the parentheses means that to count atoms, you must multiply that subscript by the numbers of atoms inside. Parentheses are useless in a chemical formula if they don't have a subscript, so we'll assume one is always there. Usually this has some meaning about the structure of the molecule, but don't worry about that for now. Very often in chemical formulae, we use parentheses to form subgroups of atoms within a molecule. This is just to show that we don't always use such simple formulae. You need a lot more chemical knowledge to interpret such a diagram, so don't worry about it for now. Carbon atoms are implied at every line end and vertex, and in such a figure, it is implied that each carbon is bonded to as many hydrogens as possible. This is an example of how a simple chemical formula like C 27H 46O doesn't really give us much of a picture of how cholesterol looks.Ī better representation of cholesterol is the shorthand stick figure below. Don't worry about these details for now just focus on counting atoms.įinally, a molecule of cholesterol contains 27 carbon atoms, 46 hydrogens and a single oxygen. Also bonded to that carbon is an OH (or "hydroxyl") group and a double-bonded oxygen. In this case, there is a CH 3, or "methyl group" bonded to a central carbon. The reason why acetic acid is often written this way is to convey a little bit of structural information – information about how the atoms are arranged in a molecule of the compound. We follow exactly the same rules for counting atoms in these kinds of formulae, we just need to add up all instances of a given atom. There are some good reasons for this that you'll learn in time. 
Some formulas contain more than one instance of a single element. It's one of the more commonly-used acids in research and industry. Sulfuric acid is a strong acid you'll learn about in the section on acids and bases. Methane is a major component in natural gas. 
Methane is one of the simplest hydrocarbons (a compound containing only carbon and hydrogen). We almost never refer to water as "dihydrogen oxide." While there are plenty of naming rules in the field of chemistry (they fill books), many chemical compounds like water and many of the ones to follow have common names. Note also that if no subscript is written, we assume it to be one. In terms of atoms, water always has a 2:1 ratio of hydrogen to oxygen. It indicates that each water molecule contains two hydrogen atoms and one oxygen. The familiar formula of water is a good place to start. We use subscripts in chemical formulae to indicate the number of atoms of an element present in am molecule or formula unit.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
